NCERT Intext Questions
1. How would you determine the standard electrode potential of the system Mg2+ ∣ Mg?
Ans. Set up an electrochemical cell consisting of Mg ∣ MgSO4 (l M) as one electrode by dipping a magnesium rod in 1 M MgSO4 solution and standard hydrogen electrode Pt, H2 (l atm) ∣ H (1 M) as the second electrode as shown in given figure.
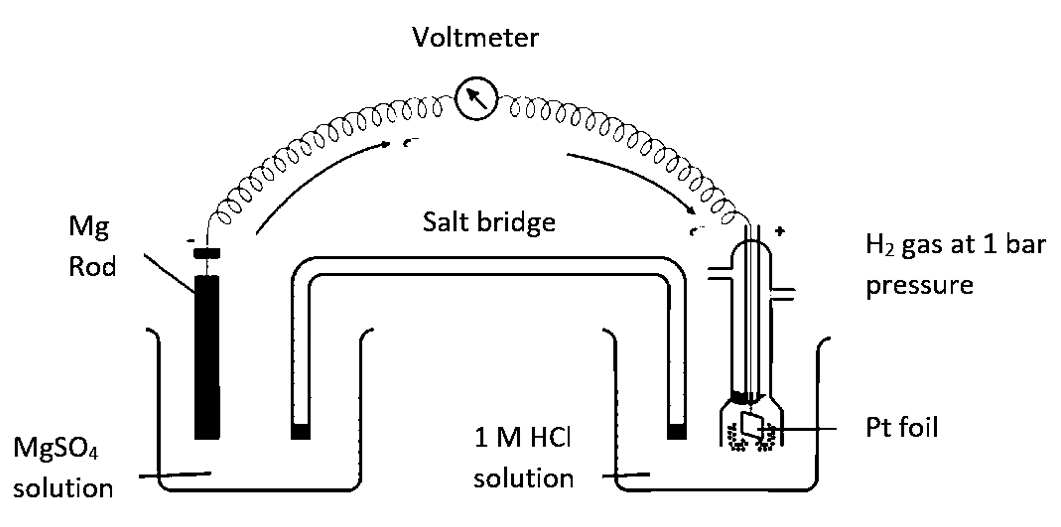
Measure the EMF of the cell and also note the direction of deflection in the voltmeter. The direction of deflection shows that electrons flow from magnesium electrode to hydrogen electrode. Thus, the cell may
Mg ∣Mg2+(1 M)∣∣ H+(1 M)∣H2(1 atm), Pt(s)
![]()
![]() = 0
= 0
![]()
2. Can you store Copper sulphate solutions in a zinc pot?
Ans. For this we have to Check whether the following reaction will take place or not.
Zn(s) + CuS04(aq) → ZnS04(aq) + cu(s)
![]() = 0.34 – ( – 0.76) = 1.10 V
= 0.34 – ( – 0.76) = 1.10 V
As ![]() is positive, the reaction will take place. Therefore, we cannot store copper sulphate in zinc pot.
is positive, the reaction will take place. Therefore, we cannot store copper sulphate in zinc pot.
3. Consult the table of the standard electrode potential and suggest three substances that can oxidise ferrous ions under suitable conditions.
Ans. Oxidation of ferrous ions means

![]() = 0.77 V
= 0.77 V
Only those substances can oxidise Fe2+ to Fe3+ which are stronger oxidising agents and have positive reduction potentials greater than 0.77 V so that Ecell of the cell reaction is positive. This is for elements lying below Fe3+ /Fe2+ in the electrochemical series, for example, Br2, Cl2 and F2.
4. Calculate the potential of hydrogen electrode in contact with a solution whose pH is 10.
Ans. For hydrogen electrode

![]()
![]()
![]() V
V
5. Calculate the emf of the cell in which the following reaction takes place:
![]()
![]()
![]()
![]() Ni(s) + 2Ag+(O.002 M) → Ni2+ (0.160 M) + 2Ag(s)
Ni(s) + 2Ag+(O.002 M) → Ni2+ (0.160 M) + 2Ag(s)
Ans. From Nernst equation to the given cell reaction
![]()
= ![]()
= ![]()
= ![]()
= ![]() = 0.91 V
= 0.91 V
6. The cell in which the following reaction occurs:
![]()
has ![]() = 0.236 V at 298 K. Calculate the standard Gibbs energy and equilibrium constant of the cell reaction.
= 0.236 V at 298 K. Calculate the standard Gibbs energy and equilibrium constant of the cell reaction.
Ans
![]() or
or ![]()
Thus, for the given cell reaction, n = 2
![]()
= ![]()
= ![]()
= ![]()
![]()
![]()
= ![]()
= – 7.983
Kc = Antilog(7.983) = 9.616×107
7. Why does the conductivity of a solution decrease with dilution?
Ans. Conductivity of a solution is the conductance of ions present in a unit volume of the solution. On dilution, the number of ions per unit volume decreases. So, the conductivity also decreases.
8. Suggest a way to determine the ![]() value of water.
value of water.
Ans ![]()
= ![]()
![]()
Thus, the molar conductivity of water at infinite dilution can be determined from the knowledge of ![]() ,
, ![]() and
and ![]()
9. The molar conductivity of 0.025 mol L-1 methanoic acid is 46.1 S cm2 mol-1. Calculate its degree of dissociation and dissociation constant.
[Given ![]() = 349.6 S cm2 mol-I and
= 349.6 S cm2 mol-I and ![]() = 54.6 S cm2 mol-I.]
= 54.6 S cm2 mol-I.]
Ans. ![]() = 349.6 S cm2 mol-I + 54.6 S cm2 mol-1
= 349.6 S cm2 mol-I + 54.6 S cm2 mol-1
![]() = 404.2 S cm2 mol-1
= 404.2 S cm2 mol-1
![]() = 46.1 S cm2 mol-1 (given)
= 46.1 S cm2 mol-1 (given)
![]()
HCOOH ⇌ HCOO– + H+
C mol L-1 0 0
c(1 – α) cα cα
![]() =
= ![]()
= ![]()
= 3.67×10-4 mol/l
10. If a current of 0.5 ampere flows through a metallic wire for 2 hours, then how many electrons would flow through the wire?
Ans. Q (coulomb) = I (ampere) x t (s) = (0.5 ampere) (2 x 60 x 60 s) = 3600 C
![]()
![]() A flow of 96500 C is equivalent to the flow of 1 mole of electrons, i.e., 6.02 x 1023 electrons
A flow of 96500 C is equivalent to the flow of 1 mole of electrons, i.e., 6.02 x 1023 electrons
3600C is equivalent to flow of electrons =![]() = 2.246 x 1022 electrons
= 2.246 x 1022 electrons
11. Suggest a list of metals that are extracted electrolytically.
Ans. Na, Ca, Mg and Al.
12. Consider the reaction:
![]()
What is the quantity of electricity in coulombs needed to reduce 1 mol of ![]() ?
?
Ans. From the given reaction, 1 mol of ![]() ions require
ions require
6F = 6 x 96500 C = 579000 C of electricity for reduction to Cr3+ ion.
13. Write the Chemistry of recharging the lead storage battery, highlighting all the materials that are involved-during recharging.
Ans. During recharging, electrical energy is supplied to the cell from an external source. The reactions are reverse of those that takes place during discharge.
PbSO4(s) + 2e– → Pb(s) + SO42- (aq)
PbSO4 (s) + 2H2O(l) → PbO2(s) + SO42- (aq) + 4H+(aq) + 2e–
2PbSO4(s) + 2H2O(l)→ Pb (s) + PbO2(s) + 4H+(aq) + SO42- (aq)
14. Suggest two materials other than hydrogen that can be used as fuels in fuel cells.
Ans. Methyl alcohol and methane.
15. Explain how rusting of iron is envisaged as setting up of an electrochemical cell.
Ans. The water layer present on the surface of iron dissolves acidic oxides of air like C02 to form acids which dissociate to give H+ ions.
![]()
![]()
In the presence of H+ ions, iron starts losing electrons at some spot to form ferrous ions. Hence, this spot acts as the anode:
Fe(s)→ Fe2+ (aq) + 2e–
The electrons thus released move through the metal to reach another spot where H+ ions and the dissolved oxygen gain these electrons and reduction reaction takes place. Hence, this spot acts as the cathode:
02(g) + 4H+ (aq) + 4e– →2H2O(l)
Over all reaction is given as
Fe(s) + O2(g) + 4H+(aq)→Fe2+ (aq) + 2H2O(l)
Therefore, an electrochemical cell is set up on the surface.
Ferrous ions are further oxidised by the atmospheric oxygen to ferric ions which combine with water molecules to form hydrated ferric oxide, Fe2O3.xH2O, which is rust.

